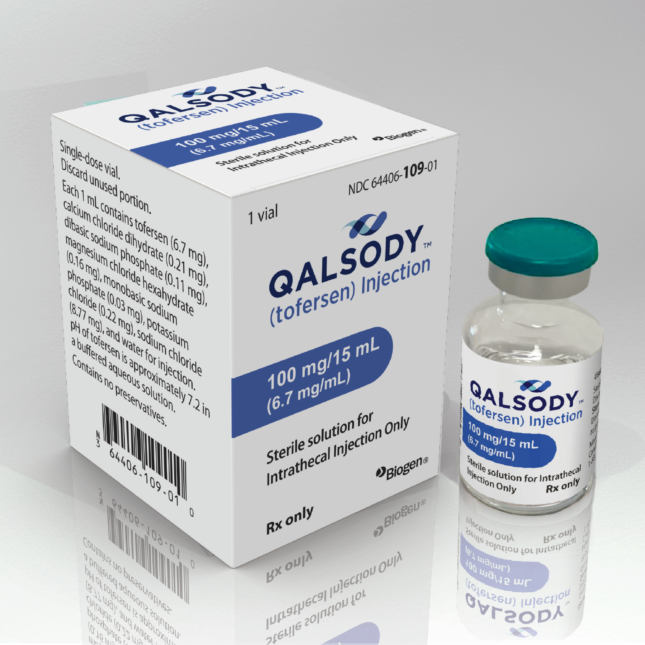
LONDON — Neurologists and patient advocates are up in arms over a policy decision by a U.K. health agency that they say will imperil access to an ALS treatment that’s available in the U.S. and on its way to approval in the European Union.
The medicine in question, Biogen’s tofersen, is designed to treat a rare genetic form of the neurodegenerative disease, accounting for some 2% of all ALS cases. And the issue is how the U.K.’s cost-effectiveness agency has said it would assess the costs and benefits of the therapy.
The agency, the National Institute for Health and Care Excellence, or NICE, has a special pathway for drugs for very rare diseases, which advocates and doctors argue is the route through which tofersen should be reviewed. But NICE has indicated it would review the drug through its standard process, saying it’s because ALS as a whole does not count as a “very rare” disease. That sets a higher bar to clear.
This article is exclusive to STAT+ subscribers
Unlock this article — plus daily coverage and analysis of the biotech sector — by subscribing to STAT+.
Already have an account? Log in
Already have an account? Log in
To submit a correction request, please visit our Contact Us page.











STAT encourages you to share your voice. We welcome your commentary, criticism, and expertise on our subscriber-only platform, STAT+ Connect