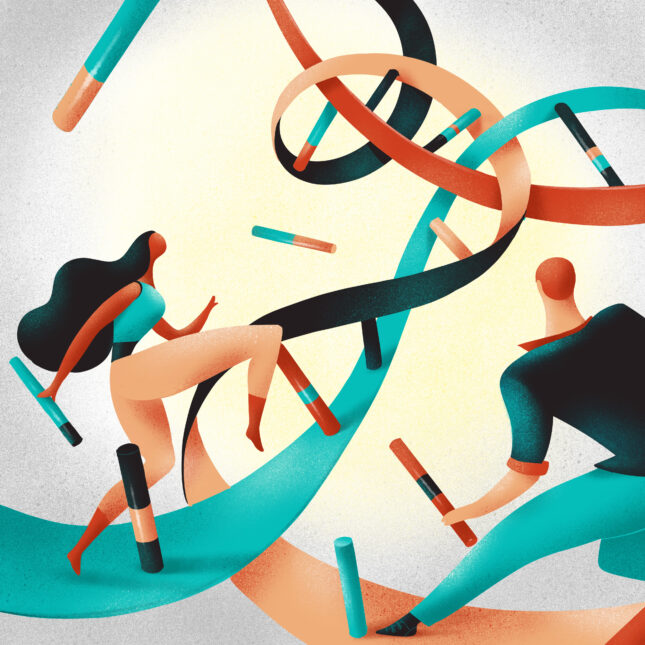
LONDON — Widespread genetic testing as well as the development of cutting-edge, customized genetic therapies have opened the door to treating many more inherited conditions than previously possible. A few recent cases, in which bespoke medicines were created for children with exceedingly rare mutations, have raised hopes for furthering the approach.
Now, researchers and government agencies in the U.K. are trying to figure out the nitty-gritty to replicate those early one-off successes for as many patients as possible — what sort of preclinical testing a tailor-made treatment needs to go through before being used, what regulatory thresholds each treatment needs to clear, and how to pay for these therapies.
“A lot of the barriers are no longer scientific — we can make these drugs,” said Matthew Wood, a professor of pediatrics at the University of Oxford. “It’s more about, can we make them at a cost that is reasonable enough? And can we get the regulators to reduce the barriers to doing this so that when a patient shows up, you can actually make a drug quickly enough to benefit that patient, rather than it costing huge amounts of money and taking two or three years to do it?”
This article is exclusive to STAT+ subscribers
Unlock this article — plus in-depth analysis, newsletters, premium events, and networking platform access.
Already have an account? Log in
Already have an account? Log in
To submit a correction request, please visit our Contact Us page.











STAT encourages you to share your voice. We welcome your commentary, criticism, and expertise on our subscriber-only platform, STAT+ Connect